NetraAI represents a distinctive, Good Clinical Practice (GCP)-authenticated machine learning architecture that offers an accessible interface for researchers to engage with consolidated multimodal datasets. This empowers sponsors to uncover explainable patient subgroups pertaining to efficacy, toxicity, and placebo response across an array of therapeutic domains, such as oncology, psychiatric disorders, neurodegenerative disorders, and rare diseases. The innovative NetraMark algorithms enable the employment of Large Language Models (LLMs), including the likes of ChatGPT, to absorb the potent hypotheses generated by the NetraAI platform from sponsor provided clinical trial patient populations, thereby enhancing the articulation of findings with increased explainability and context. NetraAI essentially functions as a data microscope, amplifying vital patient population data, and enabling new discoveries.
The true strength of the NetraAI architecture stems from its capability to discern distinct combinations of variables defining patient subpopulations, in correlation to drug or placebo response and adverse reactions—elements of key importance in clinical trials. The output of NetraAI comprises hypotheses embodied as a collection of variables and subpopulations, bolstering biostatistics, as well as supplying data to train universally applicable models. We have crafted a system whereby the output from NetraAI is fed into generative pre-trained (GPT) models using LLM APIs. Specifically, the integration of the ChatGPT API with NetraAI amplifies our capacity to generate reports that deliver clear, succinct insights to sponsors, thereby facilitating informed decision-making for their clinical trials. This novel NetraGPT module is in alignment with our mission to offer augmented intelligence algorithms capable of learning from clinical trial data, in the face of inherent challenges related to these smaller and diverse datasets.
Amplifying NetraAI with Large Language Models
Prior to recent advancements, NetraAI endowed the NetraMark team with the capacity to effectively generate reports through its unique AI-driven hypothesis-generation algorithms. These reports presented definitions of patient subpopulations via sets of variables, interpreted by NetraMark’s skilled bioinformatics team. This process enabled us to deliver valuable, human-interpreted insights to our sponsors.
Presently, Large Language Models (LLMs) undergo training with an enormous quantity of data – with models like ChatGPT4 being trained on a staggering 300 billion words of data, including medical literature. This scenario creates an unprecedented opportunity for NetraMark to seamlessly integrate outputs from NetraAI and convey potent insights about patient populations to our clientele with heightened efficiency and explainability.
Introducing NetraGPT
The NetraGPT module ingests information about specific clinical trial patient subpopulations discovered by NetraAI. More specifically, this involves feeding the collection of variables, biological or otherwise, that are statistically significant for the discovered subpopulations of interest, based on the dependent variable, e.g., drug response, placebo response, placebo non-response, adverse event, etc. The resulting output of the NetraGPT module is an easily comprehensible, detailed report, honed by the power of Large Language Models and a vast corpus of medical training, offering:
-
-
The delivery of output reports that begin to assemble in seconds and conclude within minutes
-
Enhanced explainability of the influential variables underlying the subpopulations that NetraAI discovers
-
Additional insights delineating interconnections among variables, the response, and the disease state
-
References sourced from relevant and contemporary literature.
-
In this manner, NetraAI’s remarkable ability to generate hypotheses about unknown patient subpopulations within clinical trials is amplified and expedited to deliver pivotal information to sponsors, thereby enabling them to make informed decisions concerning clinical trial enrichment and risk mitigation. By leveraging the LLM’s extensive corpus of medical training, the proprietary output of NetraAI is augmented and delivered with heightened clarity.
Critically, our process maintains transparency about the variables underpinning specific patient subpopulations, enabling clear identification and validation via statistical inspection. This level of transparency is crucial for sponsors, especially when collaborating with Large Language Models, as it permits human experts and trialists to scrutinize segments of the hypothesis-generation artificial intelligence process and the generated report, ensuring the validity of the insights and recommendations produced.
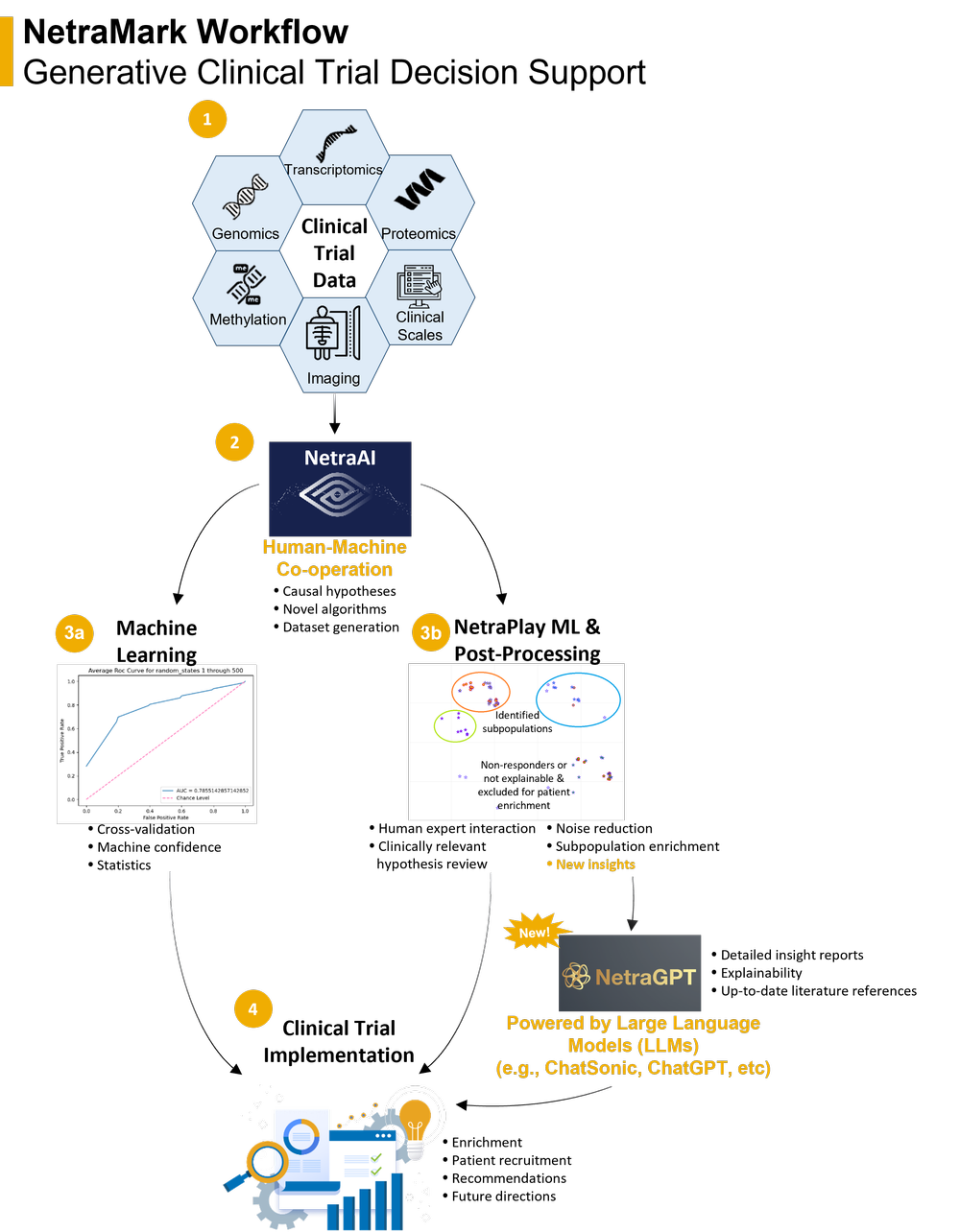
Clinical Trial Applications of NetraAI
NetraAI serves as a remarkable engine for gleaning insights about patient populations, designed to augment decision-making within the scientific leadership overseeing a clinical trial. For instance, such insights can be harnessed to enrich a patient population to help minimize placebo response prior to randomization. A distinctive aspect of the NetraAI output is a set of variables or a multidimensional fingerprint that aids in delineating a patient subpopulation. With the integration of the NetraGPT module, we can substantially speed up extracting insights about patient populations that directly influence the success and risk mitigation of clinical trials. The NetraAI system has been successfully utilized in collaboration with various sponsors across fields like oncology, psychiatry, and neurodegenerative diseases.
NetraGPT in a Schizophrenia Trial
A paramount issue in conducting clinical trials pertains to the dynamic response of participants to both the investigational drug and placebo over time, thereby necessitating the prioritization of data obtained directly from the ongoing study. For instance, psychiatric trials consistently grapple with a high placebo response. To address these challenges, NetraAI targets the “small data problem” inherent in clinical trial data, delivering clear insights and hypotheses for the leadership of clinical trials, an ability further bolstered by the integration of Large Language Models.
In a Schizophrenia trial, it was observed that certain characteristics, including a lower supine breathing rate and a higher degree of disorientation, amongst other variables, influenced the placebo response. NetraAI generated a hypothesis centered around the breathing rate and disorientation for approximately 50% of the placebo responders, and NetraGPT produced the following output:
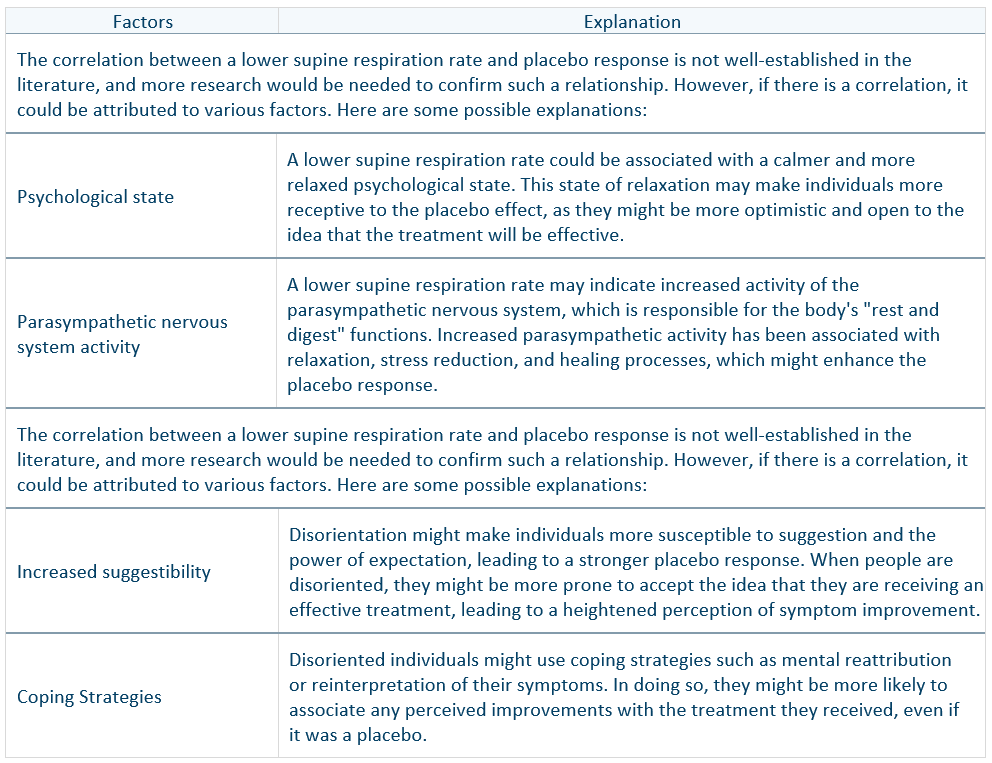
In this example, NetraAI discovered a key insight about the patient population that was tested through cross-validation, statistics, and human interpretation. The integration of LLMs, however, provided a deeper rational and explanation that warrants expert consideration. This process amplified a signal that would be difficult to detect using traditional ML methods, resulting in a comprehensive and comprehensible report that a field expert can carefully review.
NetraGPT’s Application in Oncology
Cancer comprises a complex and heterogeneous assortment of disorders, each involving a range of molecular mechanisms. NetraAI’s divide-and-conquer strategy, coupled with its foundation in systems biology, facilitates a more profound comprehension of the driving forces underlying the diverse manifestations of these diseases.
For instance, glioma (a form of brain cancer) can present in various ways in patients. Despite training the model on merely two class labels, NetraAI successfully unearthed several subpopulations (Figure 1). The interactive system of NetraPlay, equipped with a looping tool, enables users to delve into patient subpopulations and extract output from statistical tests that evaluate the significance of the discovery.

Figure 1. NetraAI perspective of the different glioma patient subpopulations. Using NetraPlay, a subpopulation of interest is looped and variables driving that subpopulation are revealed.
Predicated on the variables influencing the chosen subpopulation, NetraGPT can produce a report (Figure 2), aiding users in identifying enrichment criteria for prospective trials. Importantly, the depth and framework of the report can be tailored to specific requirements and is constantly evolving.
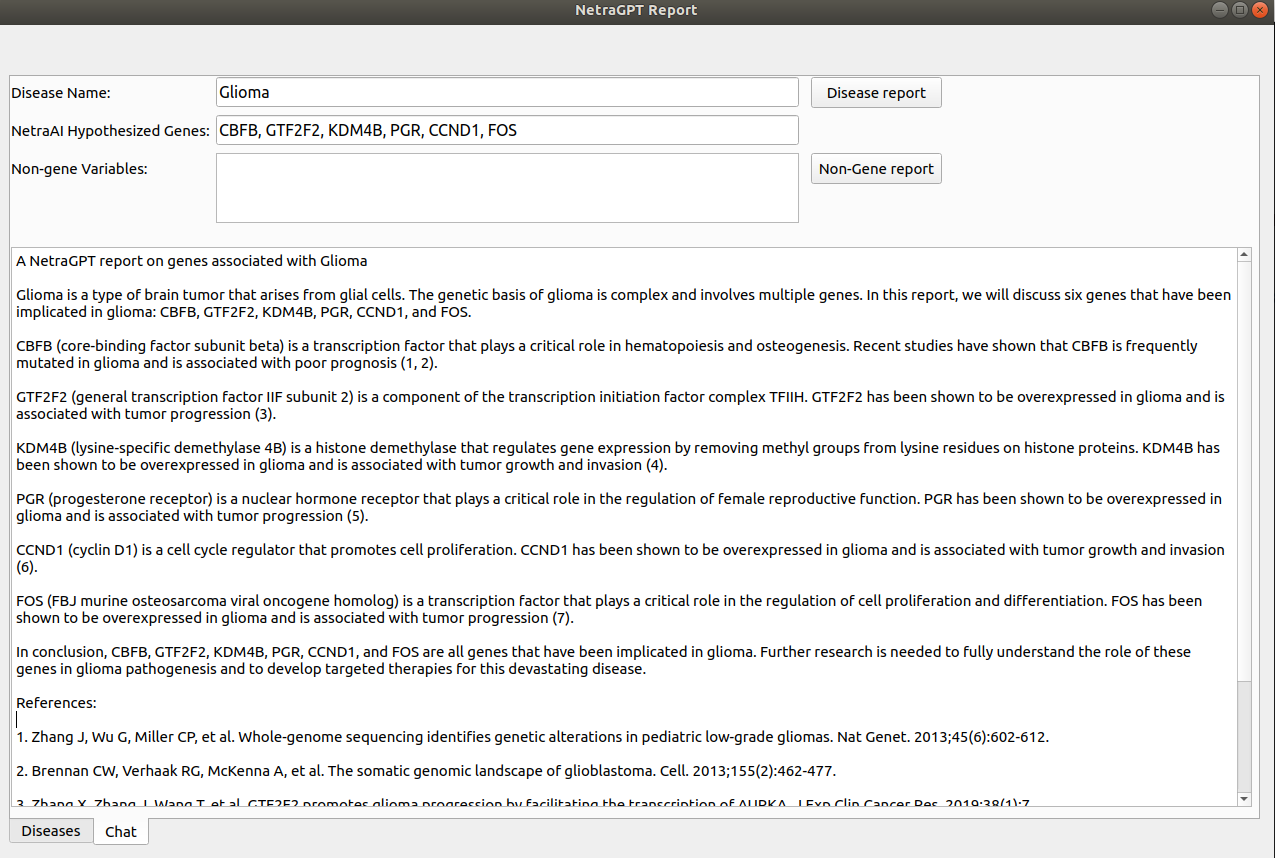
Figure 2. NetraGPT report for a subpopulation of glioma identified using NetraAI. A report is generated using the driving variables for the specific subpopulation.
Impact of NetraGPT
The amalgamation of Large Language Models (LLMs) with NetraAI, resulting in the development of the NetraGPT module, has elevated our system’s capabilities, keeping the requirements of our clientele at the forefront. While NetraAI has always demonstrated proficiency in distilling crucial insights from compact clinical trial datasets for expert evaluation, the NetraGPT module enhances the delivery of these valuable insights to sponsors with greater efficiency. Harnessing cutting-edge technology, we aspire to continuously transform the landscape of clinical trial enrichment and risk mitigation, thereby expediting the introduction of secure and efficacious drugs to the market, all the while maintaining paramount transparency and explainability.

